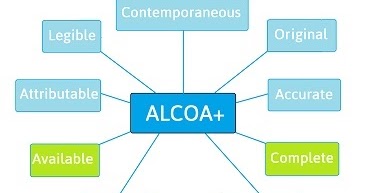
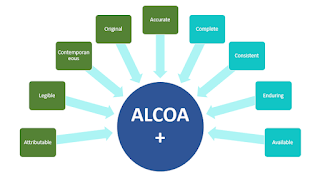
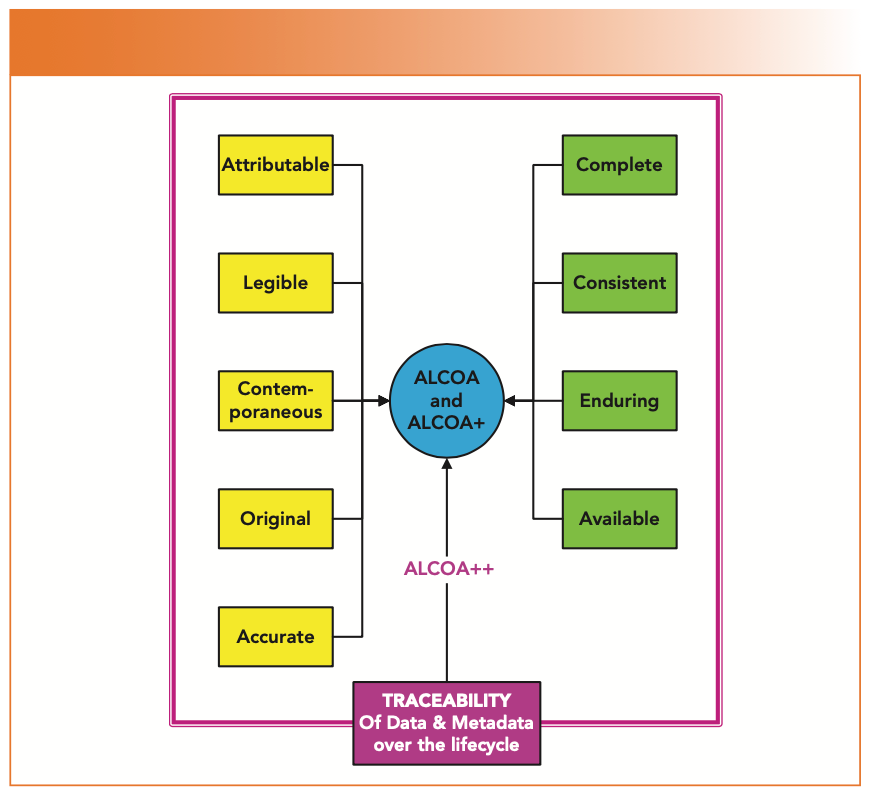
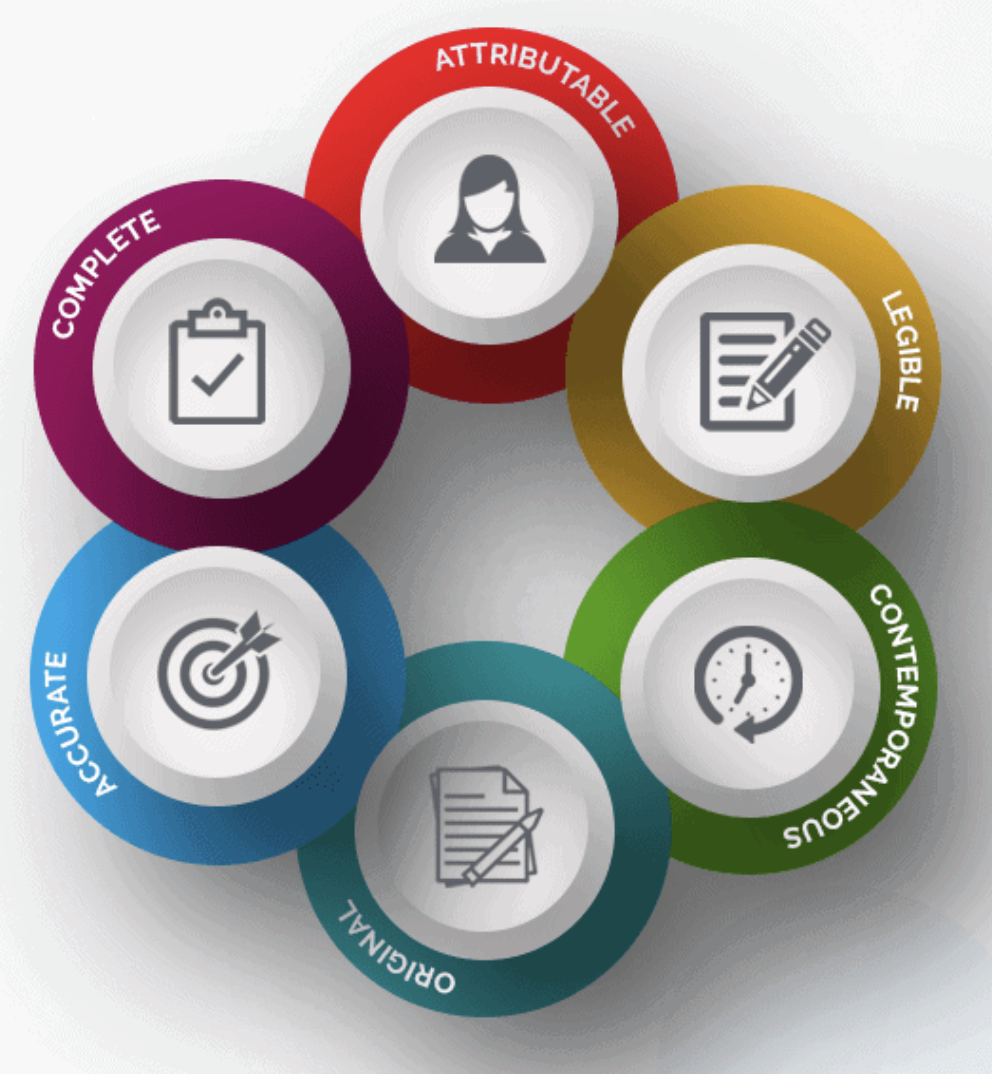
New Life Clinical Research on Twitter: "#ALCOA + C to achieve #data quality. #NewLifeClinicalResearch #nlcr #NewLifeResearch #NLCR #nlcresearch #alcoac https://t.co/RWUMbEuuvy" / Twitter

Certified Copies and ALCOA-C: Essentials Post ICH GCP E6 (R2) Addendum - Life Science Training Institute