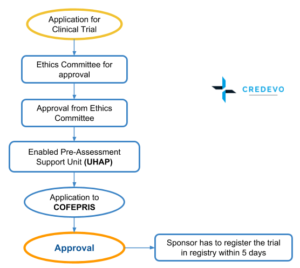
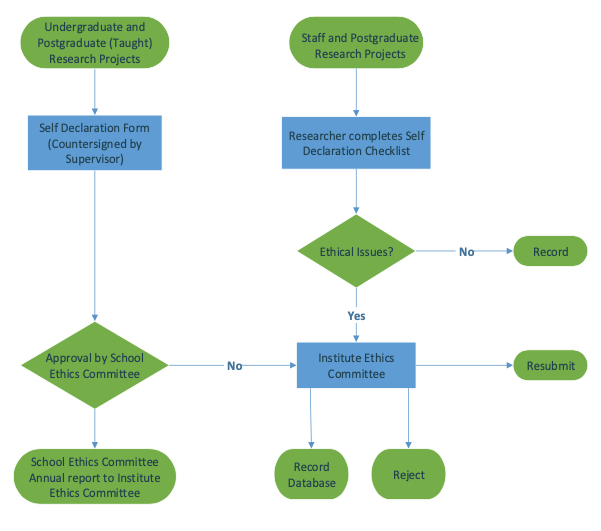
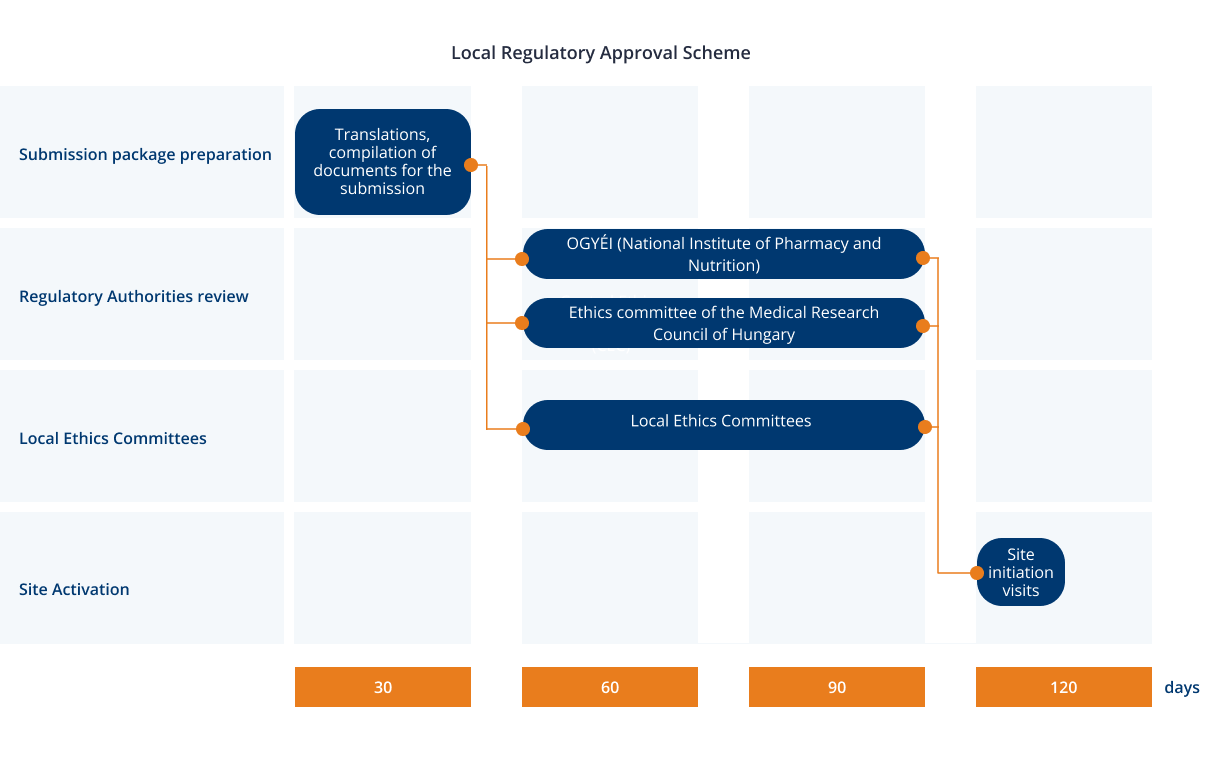
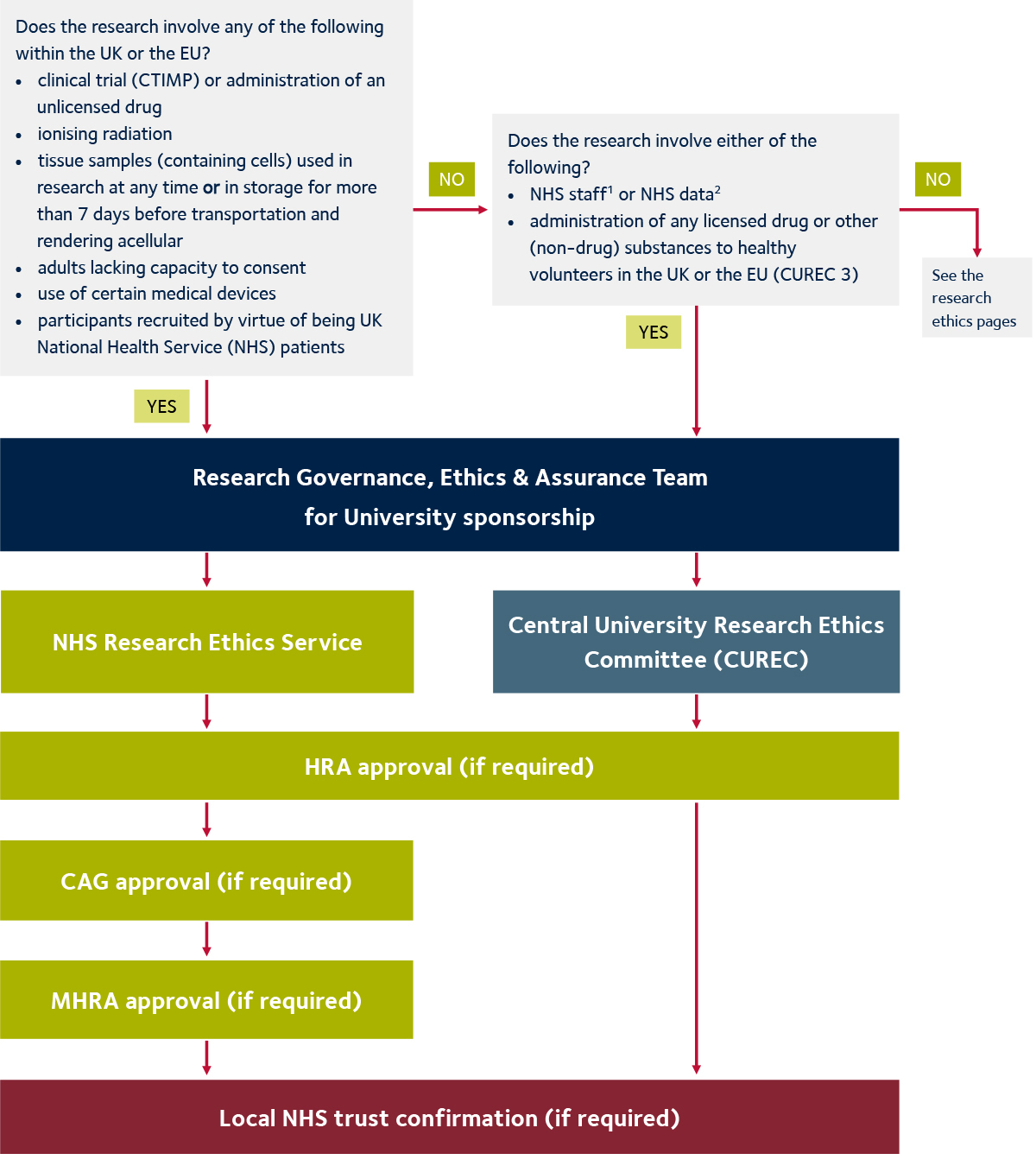
Vulnerable population and methods for their safeguard – topic of research paper in Clinical medicine. Download scholarly article PDF and read for free on CyberLeninka open science hub.
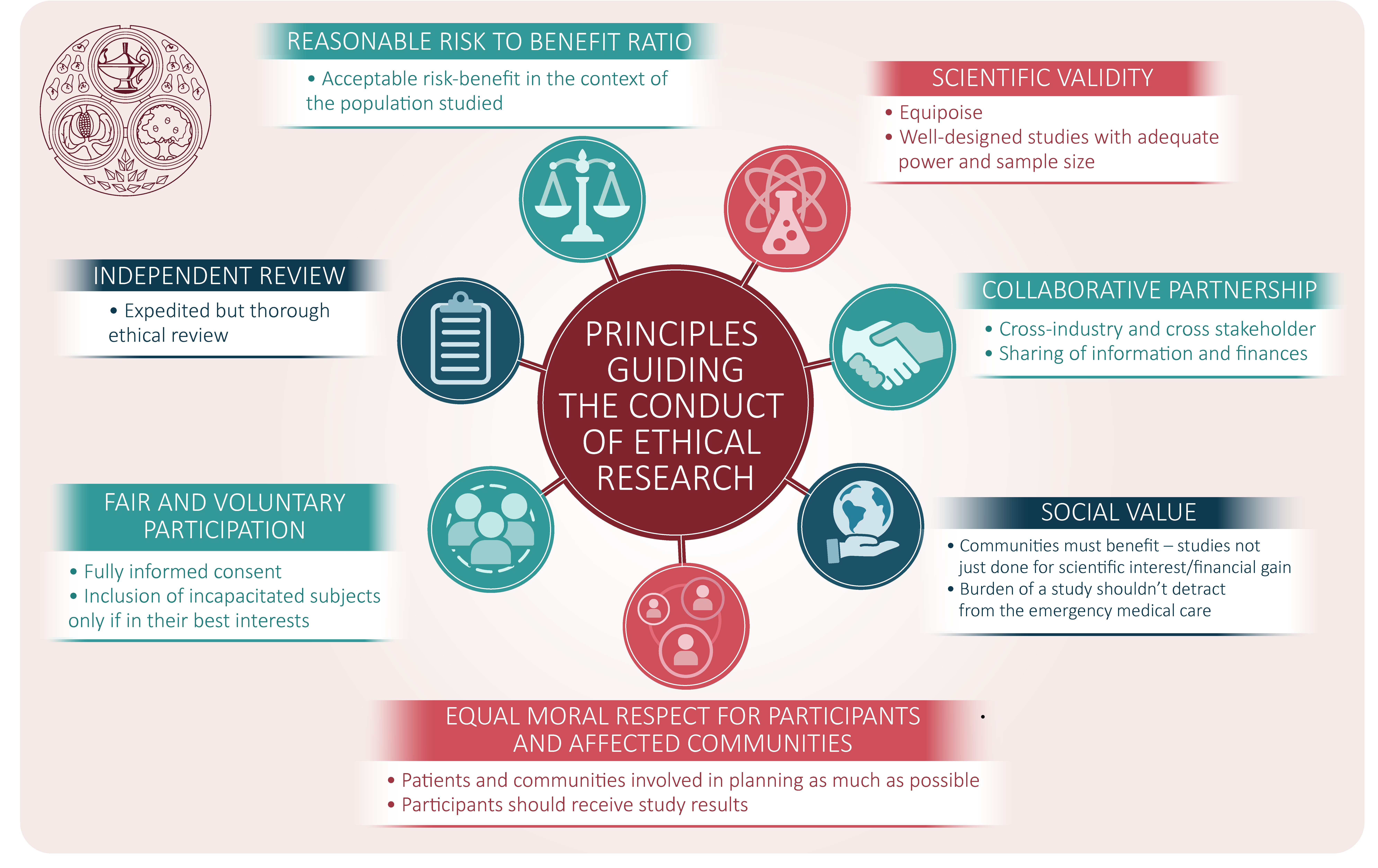
The ethics of conducting clinical trials in the search for treatments and vaccines against COVID-19 - FPM