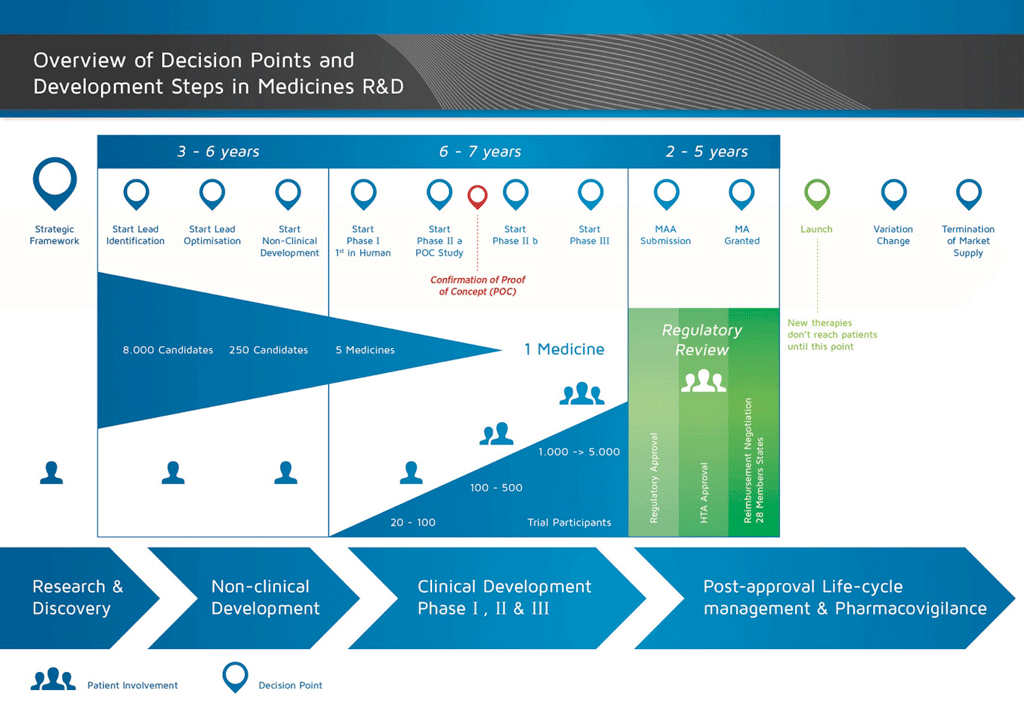
Steps of non-clinical studies in drug development process. GLP: Good... | Download Scientific Diagram
M3(R2) Nonclinical Safety Studies for the Conduct of Human Clinical Trials and Marketing Authorization for Pharmaceuticals
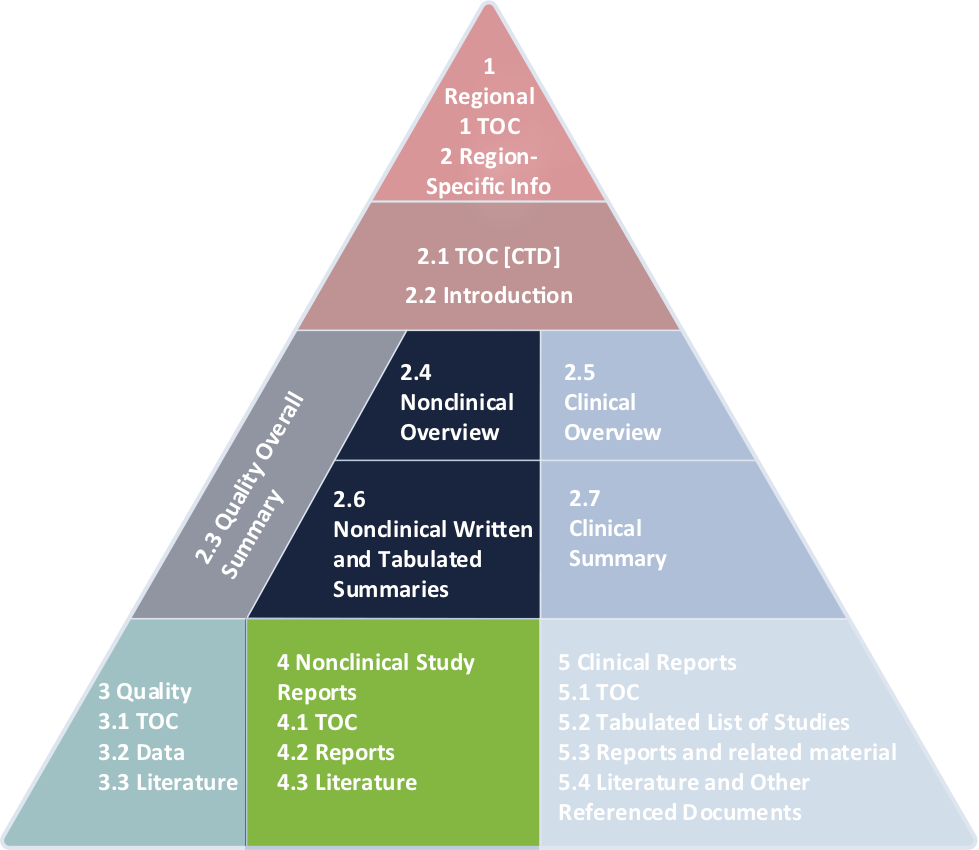
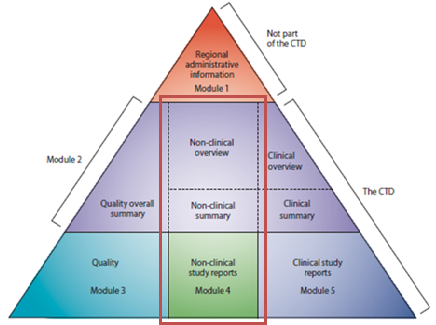
ICH M3 (R2): Non-Clinical Safety Studies for the Conduct of Human Clinical Trials and Marketing Authorization for Pharmaceutica

ICH M3(R2) - Guidance on Nonclinical Safety Studies for the Conduct of Human Clinical Trials and Marketing Authorization for Pharmaceuticals
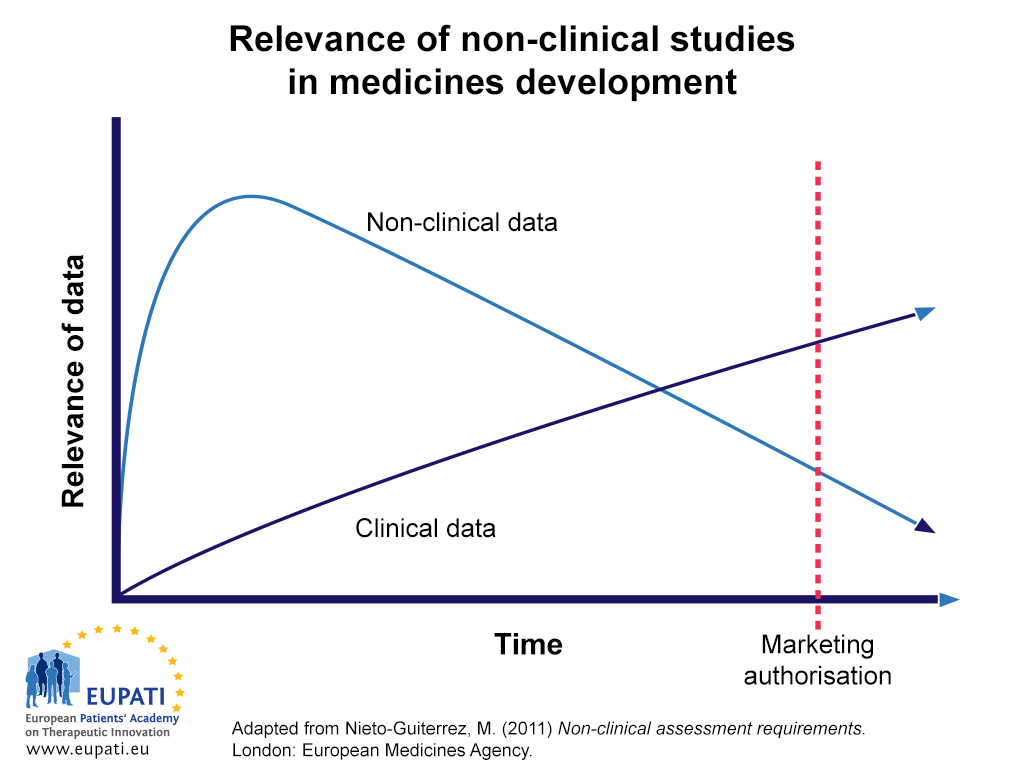
Non-clinical studies in the process of new drug Development--Part II: Good laboratory practice, metabolism, pharmacokinetics, safety and dose translation to clinical studies - Document - Gale Academic OneFile

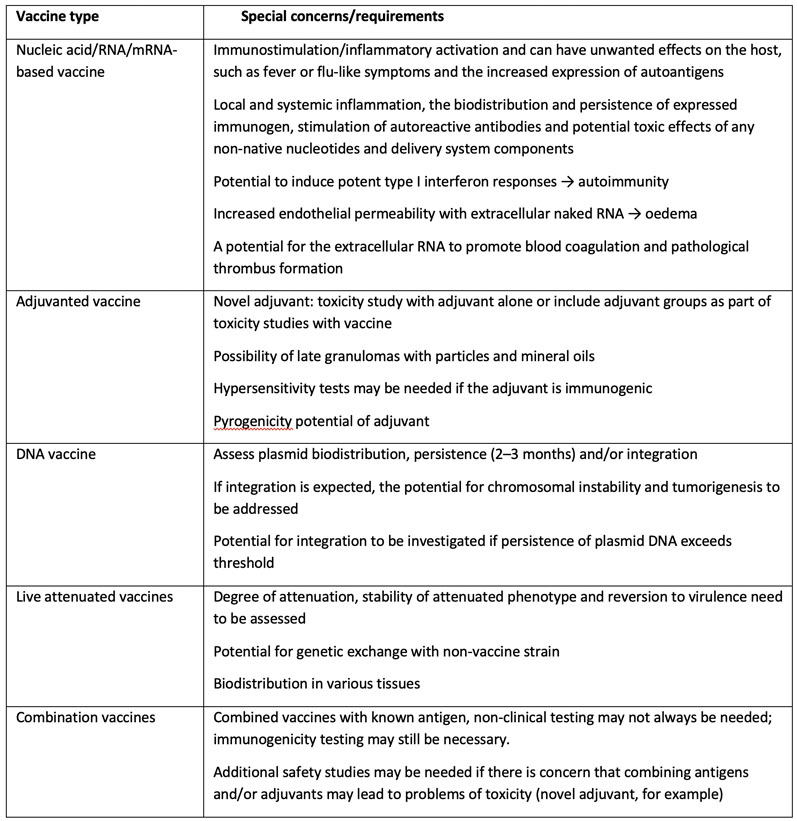
Non-clinical assessment of cell therapy products: the perspective from five Asian countries/regions based on regulatory guidelines and the underpinning rationales - Cytotherapy
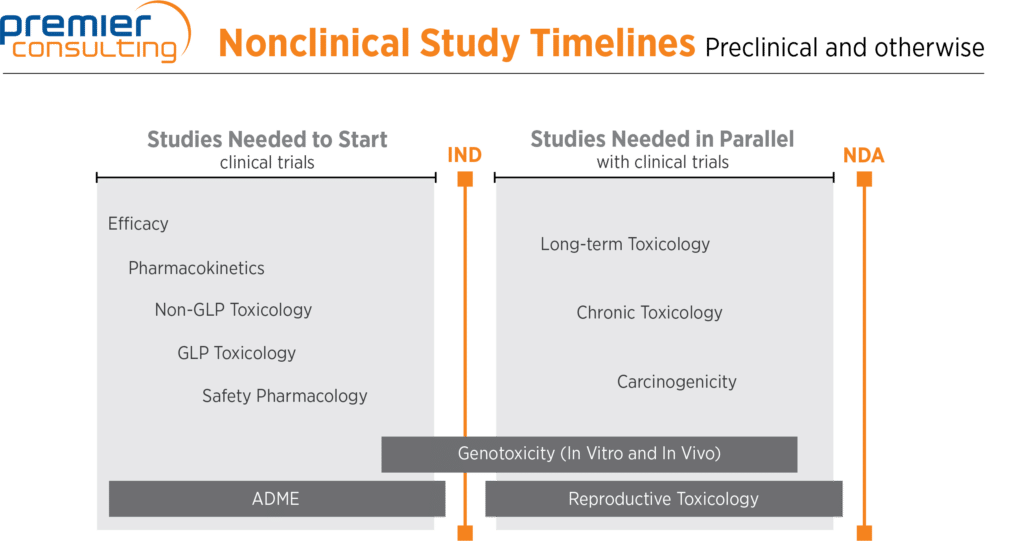
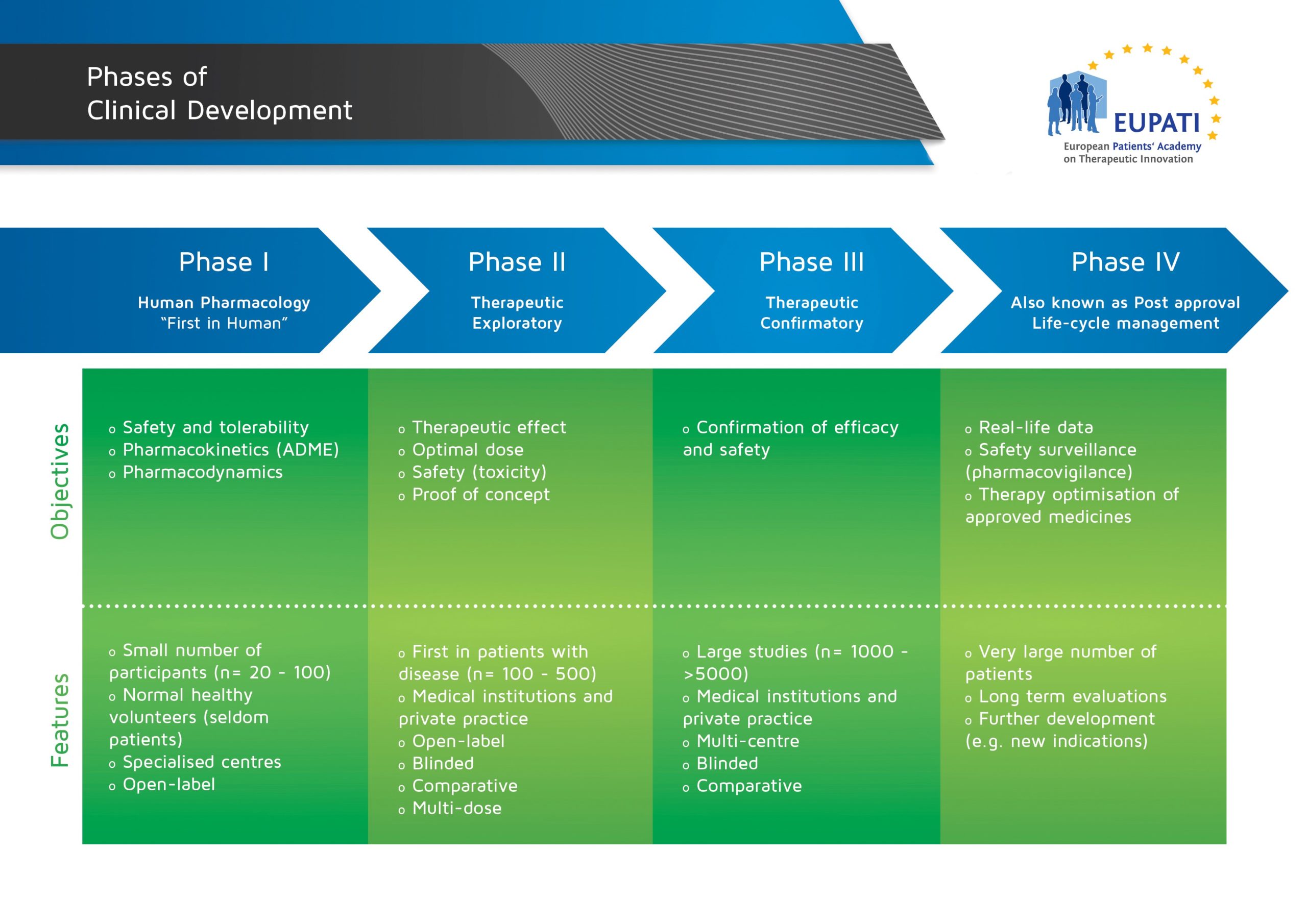
Preclinical And Nonclinical Studies—What Is The Difference, And Where In Your Program Should They Fall? | Premier Consulting
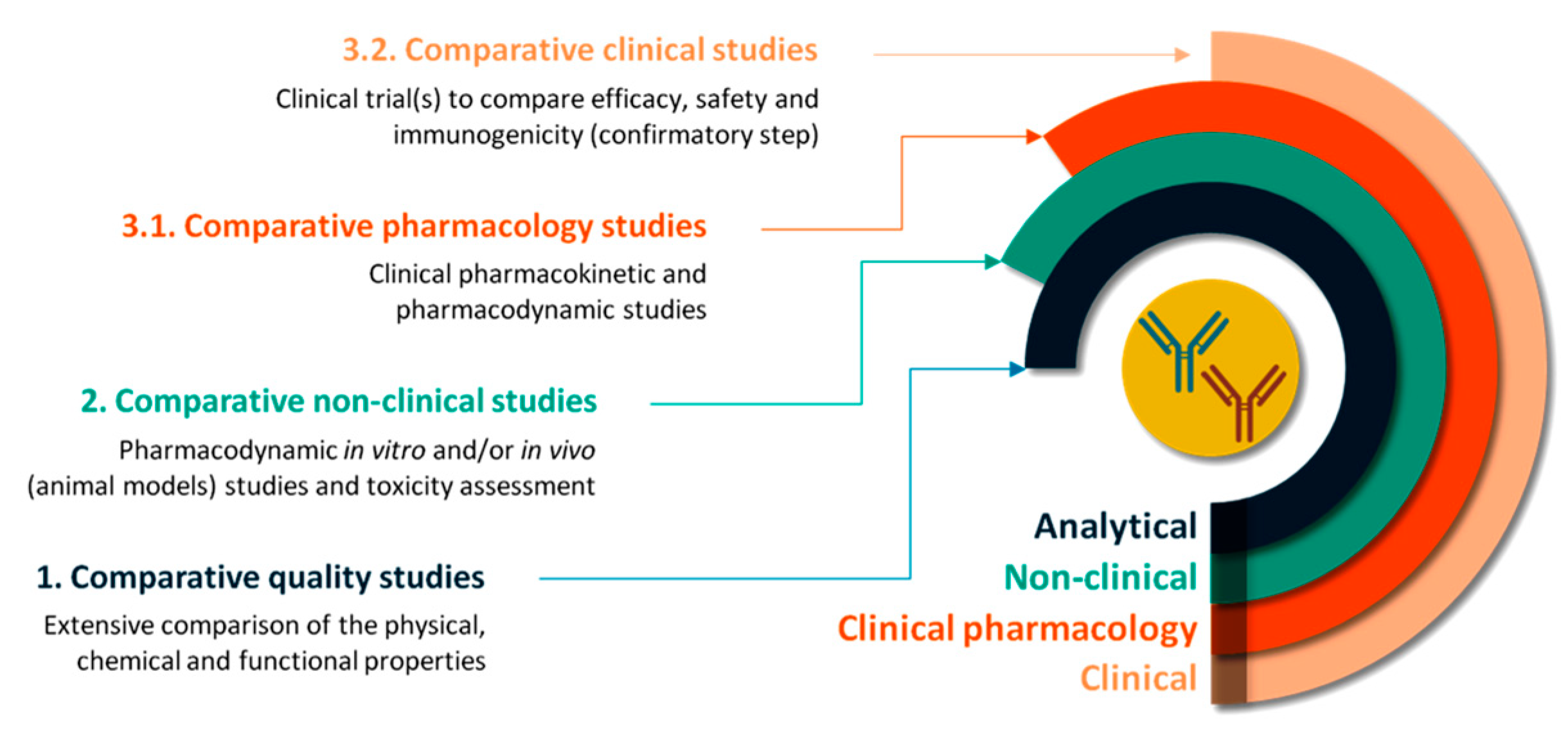
Pharmaceutics | Free Full-Text | Concepts and Challenges of Biosimilars in Breast Cancer: The Emergence of Trastuzumab Biosimilars

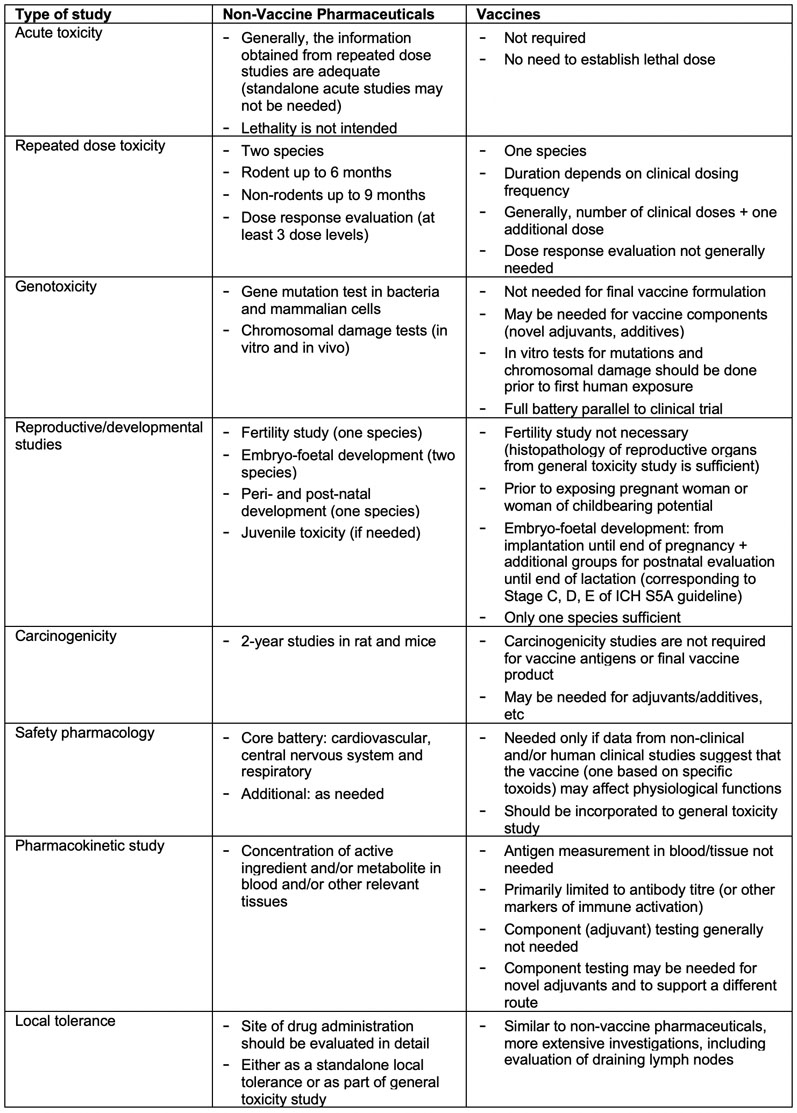
Table 1 from Non-clinical studies in the process of new drug development - Part II: Good laboratory practice, metabolism, pharmacokinetics, safety and dose translation to clinical studies | Semantic Scholar

Conclusions from non-clinical safety studies of the OPCs to be injected... | Download Scientific Diagram